MATTER
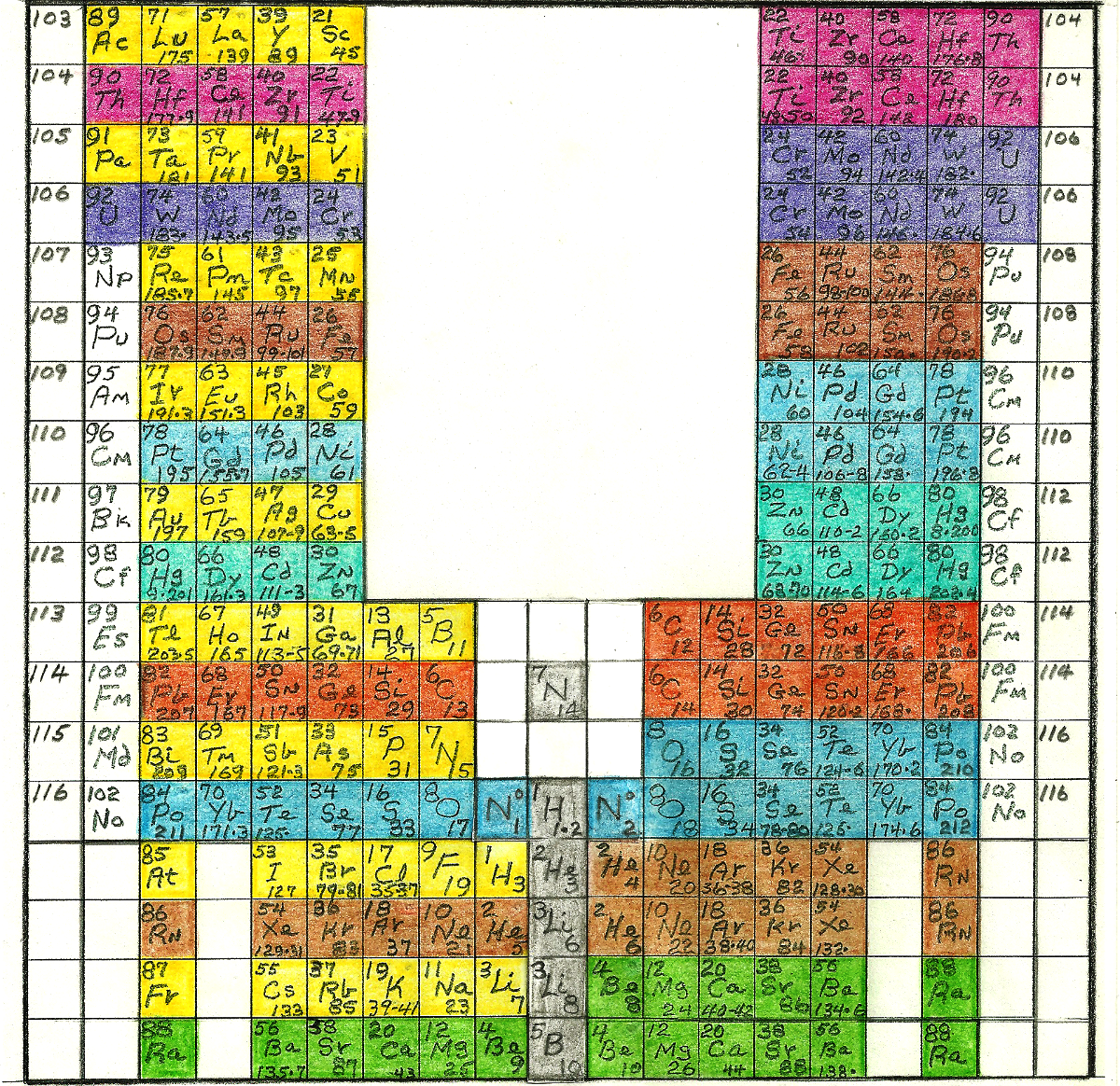
Periodic Tables of Matter. The Elements by Mass and Atomic Numbers, Orbitals and Ionic Bonds.
Table Attributes link

Pauli Exclusion Principle
Electrostatic Repulsion by the Electron’s Electric Field.
GEOLOGIC BIOLOGIC
Teaching Aids:
Periodic Table Tiles and Posters…
Hypothetically,
the Nucleus is not a bag of Marbles.
Arranged in order of Mass Numbers, in two distinct Sets, the Geologic and Biologic Series.
The Biologic Series is in two sets of High and Low Mass, B- and B+ Decay respectively.
Illustrated are the Periods, Groups and Transition Zones, after Mendelev 1850.
Also shown are:
the Integrated Rare Earth Isotopes.
the Electron Orbital Shell Periods and Groups.
the Dynamic Isotopes in Gray, adjacent their Parent Isotopes.
and the Magnetic Group.
and the Radioactive Group.
and the Lanthanides and Actinides.
All Stable Isotopes are Bounded by Neutrons.
All Stable Isotopes are Symmetrical.
All Stable Isotope have Equal number of Positrons and Negatrons.{Universal Symmetry}
No Geologic Isotopes have the S Shell filled.
All Biologic Isotopes have the S shell filled.
All Geologic Isotopes suffer Orbital Magnetic Moments.
No Biologic Isotopes suffer Orbital Magnetic Moments.
All Geologic Isotopes are Stacked NPN, with a Proton Core.
All Biologic Isotopes are Stacked NPPN, with an Alpha Core.
No Geologic Isotopes can suffer Alpha Decay by Fision.
All Biologic Isotopes can suffer Alpha Decay by Fision.
All Geologic Isotopes have Odd Mass Numbers.
All Biologic Isotopes have Even Mass Numbers
Biologic Isotopes have Atomic Field Strength two X Stronger than Geologic Isotopes, Fills the S Orbital.
These Dynamic Isotopes are Non-Symmetrical and Suffer Nuclear Magnetic Moments.
1H1 is N* by B-decay, is not Bounded by Neutrons.{Diatomic}
1H2 is is not Bounded by Neutrons.
2He2 is not Bounded by Neutrons.
2He3 is 1H3+ with a Captured Electron.{MASS 3 DEBATE}
3Li6 is 2He6 by B- Decay, not Bounded by Neutrons.
5B10 is 4Be10 by B- Decay, not Bounded by Neutrons.
7N14 is 6C14 by B- Decay, not Bounded by Neutrons.
CNO Cycle:6C12+8O16 = 14N28 =7N14 +7N14 {Diatomic}
Others not shown:
19Kp 40, 23V 50, 43Tc 98, 57La 138, 71Lu 176, 73Ta 180…
Nuclear Magnetic Moments

Teaching Aids:
GeoMag, Magnets and Field Viewers…
ORBITALS
Van Allan Model

Orbital Magnetic Moments

Ionic Bonds

Biologic Series is 2X stronger than Geologic
Requires non-averaged Isotope bonding energies for Geo and Bio series.
Links
Alt. Chemistry Link
Jefferson Lab Lecture
https://environmentalchemistry.com/yogi/periodic/
http://seyahacademy.ca/seyahs-periodic-tables18/
http://seyahacademy.ca/a-field-theory/
http://seyahacademy.ca/a-field-theory-ii/
http://seyahacademy.ca/field-theory-iii/
Seyah Periodic Tables
Not for Scardy Kittens or Baby Criers
Share
Hits: 13018
